Glycobiology of Leukocyte Adhesion, Atherosclerosis, and Thrombosis
A general overview of our laboratory’s interests:
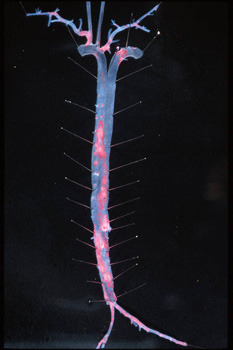 |
| Image: Aorta and great vessels from an apolipoprotein E-deficient mouse fed a high fat (western) diet for 6 months. Aorta is opened lengthwise and stained with Oil-Red-O (red) to demonstrate fat within atherosclerotic lesions. |
Our laboratory is interested in understanding the role of fucosylated glycans in the glycobiology inherent to inflammatory and/or immune cardiovascular diseases, as well as their role in hemostasis and thrombosis. The leukocyte adhesion cascade is a complex multi-step process that captures free-flowing leukocytes from the bloodstream. It includes leukocyte tethering to, rolling on and firm adhesion to endothelial cells, and transmigration across the vascular wall. Leukocyte tethering and rolling are largely dependent on selectin adhesion molecules and their ligands. Fucosylated glycoproteins that serve as ligands for the selectins are therefore critical components of the leukocyte adhesion cascade. These glycoproteins are important in the development of inflammatory and immune vascular diseases like atherosclerosis. One of the selectins, P-selectin, and its primary ligand, P-selectin glycoprotein-1 (PSGL-1), seem to serve as a link between the inflammatory response and the coagulation system. In addition to their role in leukocyte trafficking, these molecules are thought to promote thrombosis by several mechanisms.
Atherosclerosis is commonly referred to as “hardening of the arteries.” It is the underlying cause of most “heart attacks” and “strokes,” and is therefore a leading cause of death in the United States and other westernized countries. Our work in this field focuses on understanding how the selectin-type adhesion molecules and their fucosylated glycoprotein ligands contribute to the recruitment of various leukocyte types at the sites necessary for atherosclerotic disease development and progression. Particular attention is directed toward understanding the role of two enzymes, alpha(1,3)-fucosyltransferase IV and VII (FUT4 and FUT7, respectively), in the synthesis of the fucosylated glycans on selectin adhesion molecule ligands used for leukocyte homing and trafficking in the atherosclerotic disease process. Our goals are to understand 1) which types of leukocytes utilize the fucosyltransferases to synthesize the fucosylated selectin ligands necessary for the development and progression of atherosclerosis, and 2) how expression or regulation of the enzyme activity can modulate development and progression of atherosclerosis.
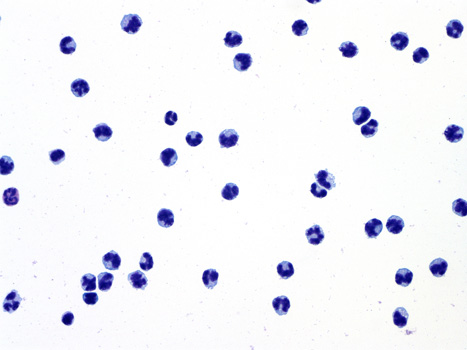 |
| Image: Murine peripheral blood monocytes purified by a negative immunoselection technique. Monocyte-derived microparticles that express Selectin ligands like PSGL-1 are thought to modulate thrombotic processes. |
Thrombosis is the pathologic formation of a blood clot within the lumen of a vessel. Heart attacks and strokes are generally caused by acute thrombosis superimposed upon atherosclerotic disease. A number of biologic links between components of the inflammatory system and the coagulation cascade have been identified recently, including modulation of the cascade by forms of P-selectin and its fucosylated ligand, PSGL-1. These links have led us to investigate the role of FUT4 and FUT7 in the production of fucosylated glycans that may modulate hemostasis and thrombosis. Our studies clearly demonstrate an unexpected thrombotic phenotype in mice lacking these fucosyltransferases. With respect to this project, our current goals are to 1) fully characterize the phenotype, 2) determine the molecular mechanisms that account for the phenotype, including the role of FUT4 and FUT7 in the synthesis of relevant glycoconjugates.