Additional Information: Parents and Infants Engaged
Participate now
Principal Investigators:
Linda Watson, EdD, The University of North Carolina at Chapel Hill
Grace Baranek, PhD, The University of Southern California
Investigators:
Elizabeth Crais, PhD
Lauren Turner Brown, PhD
Richard Faldowski, PhD
Study Goal:
To identify early signs of developmental problems in infants from 11 to 16 months of age using the First Years Inventory (FYI), and then to test an in-home parent coaching intervention that we believe will help infants who are at risk for later developmental problems such as language disorders, sensory regulation problems, and autism.
Procedures:
The study is designed as a randomized comparative trial. We are recruiting 44 infants at-risk for developmental disorders and their caregiver(s).
- We will recruit participants by mailing the FYI to families of 11-to-16-month-old infants based on public birth records, as well as distributing the FYI through area pediatric practices and public health clinics.
- Infants who are identified as being at elevated likelihood for neurodevelopmental conditions based on parent report on the FYI are asked to participate in a developmental assessment. Infants who are eligible for the study are randomly assigned (like a coin flip) to be initially coached on either the Sensory Regulatory or Prelinguistic Communication (PC) domain of PIE. Families return for assessments after the first 6 weeks of coaching, after which they will be coached in the other domain of PIE. Following completion of the 13 total weeks of coaching, infants participate in a final developmental assessment.
PIE Intervention:
The 13 in-home coaching sessions are designed to increase the amount of time infants spend in joint engagement with their parents during daily routines. Half of the intervention sessions focus on sensory regulation and half focus on pre-linguistic communication skills. Families are randomly assigned (like a coin flip) to start with one domain or the other, but all families receive coaching in both domains. Though PIE is made up of evidence-based practices, this is a “proof of concept” study, which means that the PIE intervention has never been tested before with infants at higher likelihood for developmental problems and the intervention may evolve as we learn from families in the study.
Inclusion Criteria:
- For the intervention trial: infant must (1) meet risk criteria on the FYI screener and (2) score at least one standard deviation below the mean on either the receptive or expressive language subscales of the Mullen Scales of Early Learning AND meet Sensory Processing Assessment inclusion criteria for hypo- or hyper- sensory behaviors. All families who do not meet inclusion criteria will be given information about community services that may be helpful for their child’s development.
- How we determine risk?: Risk is calculated based on data collected in another study where risk status on the FYI was confirmed with follow up at three years. We empirically determined combinations of the two FYI domain scores that would sort respondents into “at-risk” and “not-at-risk.” The resulting cut-points enabled us to sort children such that we capture about one-third of those who would go on to be confirmed as at-risk while misclassifying less than five percent of the typically developing sample as at-risk. Being identified as at-risk for this study does not mean that a child will necessarily go on to develop a neurodevelopmental condition; they are just at higher likelihood to do so than the general population.
- What are hypo-sensory behaviors?: the absence of or delayed orienting to social and nonsocial stimuli in a child’s environment (for example, not responding to their name being called or not responding to a phone ringing until it has rung several times)
- What are hyper-sensory behaviors?: over-reaction or avoidance of social and non-social stimuli in a child’s environment (for example, excess fussing when wet, crying or showing fearful responses to everyday noises).
Exclusion Criteria:
- Families who speak English less than 50 percent of the time at home.
- Infants with previously identified genetic disorders (e.g., Down syndrome).
- Infants with identified vision/hearing/physical impairments.
- We will refer any families who are ineligible for this study to community services that may be helpful for their child’s development.
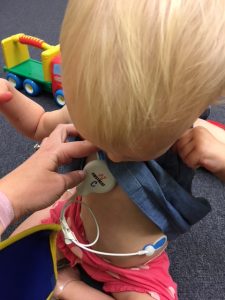
As part of the assessments for this study, we are collecting physiology (heart rate and sweat) data on children while they watch a short video and play with their parent. The parent wears chest electrodes and the child wears electrode stickers on their chest and left foot. We will help prepare children for this painless but unusual experience.


This study is funded by a Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) R21 grant awarded to Dr. Linda Watson and Dr. Grace Baranek. This is a registered clinical trial.
Contact:
Jonet Artis, Project Coordinator
Phone: (919) 843-5498
E-mail: PIEstudy@unc.edu