
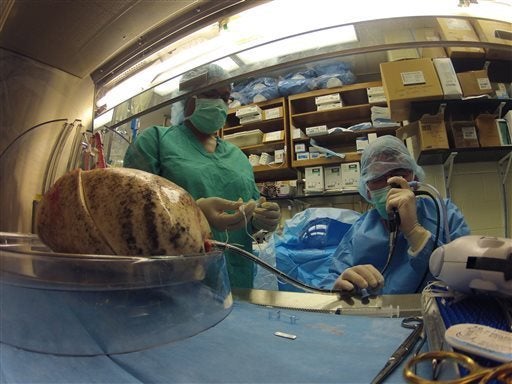
A clinical trial underway at the University of North Carolina is testing the idea that lungs can be recovered for transplant from people who die suddenly, away from the hospital. Lungs for transplant normally come from people who have suffered brain death and have been on a ventilator in a hospital, but many lungs from these donors are not usable because of infection or inflammation resulting from brain injury. Many people with end-stage lung disease are unable to get lung transplants because of the serious shortage of suitable lungs.
Thomas M. Egan, M.D., is principal investigator of the UNC study, which is funded by the National Institutes of Health/National Heart, Lung, and Blood Institute (NHLBI grant 1 UM1 HL113115-01A1). UNC co-investigators include Benjamin Haithcock, M.D., and Jason Long, M.D., of the UNC Division of Cardiothoracic Surgery; Peadar Noone, M.D., of the UNC Division of Pulmonary Diseases and Critical Care Medicine; Paul Stewart, Ph.D., of the UNC Department of Biostatistics and Katherine Birchard, M.D., of the UNC Department of Radiology.
The Associated Press reported on the study in a story on Nov. 17, 2013.
Dr. Egan pioneered the idea that lungs could be suitable for transplant for hours after circulation stops because lung cells obtain oxygen after death from air sacs and airways (other organs require blood circulation to obtain oxygen). He believes that the lungs from people who have died suddenly may not only be plentiful; they may be superior to lungs transplanted from brain-dead donors.
The study uses lungs from people in Wake County, NC, who have indicated that they wish to be organ donors and who have died of sudden cardiac arrest, usually at home. If emergency crews fail to resuscitate the person, organ donation officials talk to family members to request use of the lungs for possible transplant. If the family agrees, the body is transported to UNC Hospitals, where the lungs are recovered and assessed for transplant using ex-vivo (outside the body) perfusion. Lungs that are found acceptable will be transplanted to patients who have consented to use of the lungs under protocols approved by the Food and Drug Administration (FDA) and the UNC Institutional Review Board (IRB). Outcomes will be compared to patients who have received transplanted lungs from conventional donors. No other research team in the United States has FDA approval to test transplants of lungs from sudden-death donors.
Wake County Emergency Medical Services, law enforcement, the North Carolina medical examiner’s office, and other government offices are cooperating in the project.
Contact: Margaret Alford Cloud, UNC Division of Cardiothoracic Surgery, mcloud@med.unc.edu
