Tissue Culture and Smoke Exposure Core
Well-differentiated normal human bronchial epithelial cultures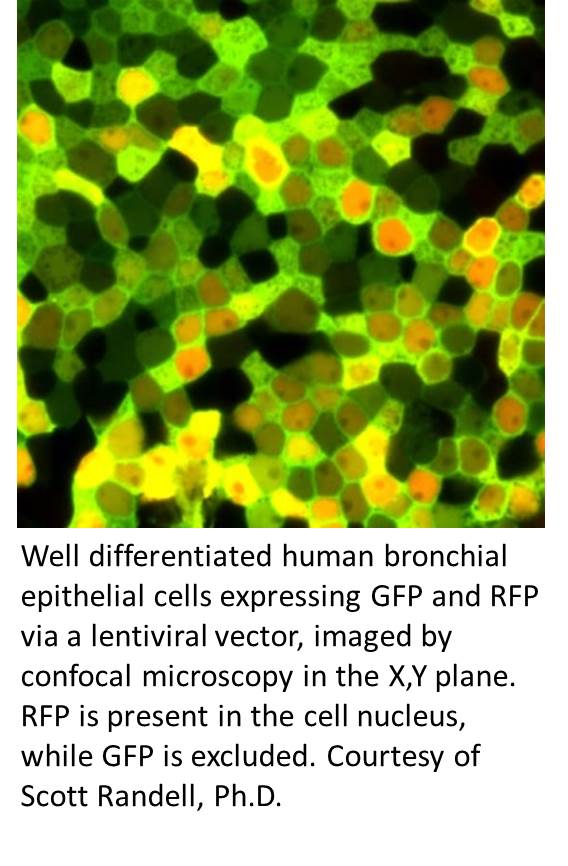 are instrumental for studying basic and applied aspects of tobacco smoke respiratory tract toxicity. Primary cells are likely more predictive than transformed cell lines and poorly differentiated primary cells grown under non-differentiated submerged conditions. Understanding this, this core will provide the following 3 services: 1) procure lung tissue for airway epithelial cell culture using long-standing procedures; 2) isolate and culture epithelial cells from excised human lungs for project investigators, including the preparation of the appropriate number of polarized well-differentiated airway epithelial cultures from passaged or cryopreserved cells on permeable substrates as dictated by project needs; and 3) carry out epithelial cell quality control (QC) by subjecting passage 2 cells from each dedicated lung and from select nasal specimens to a series of QC procedures including growth in antibiotic free media, ability to form a pseudostratified mucocilary epithelium (histology and/or whole mount immuno-staining and confocal analysis) and mycoplasma testing.
are instrumental for studying basic and applied aspects of tobacco smoke respiratory tract toxicity. Primary cells are likely more predictive than transformed cell lines and poorly differentiated primary cells grown under non-differentiated submerged conditions. Understanding this, this core will provide the following 3 services: 1) procure lung tissue for airway epithelial cell culture using long-standing procedures; 2) isolate and culture epithelial cells from excised human lungs for project investigators, including the preparation of the appropriate number of polarized well-differentiated airway epithelial cultures from passaged or cryopreserved cells on permeable substrates as dictated by project needs; and 3) carry out epithelial cell quality control (QC) by subjecting passage 2 cells from each dedicated lung and from select nasal specimens to a series of QC procedures including growth in antibiotic free media, ability to form a pseudostratified mucocilary epithelium (histology and/or whole mount immuno-staining and confocal analysis) and mycoplasma testing.
Additionally, this core will house the Vitrocell VC10 Smoking Robot and 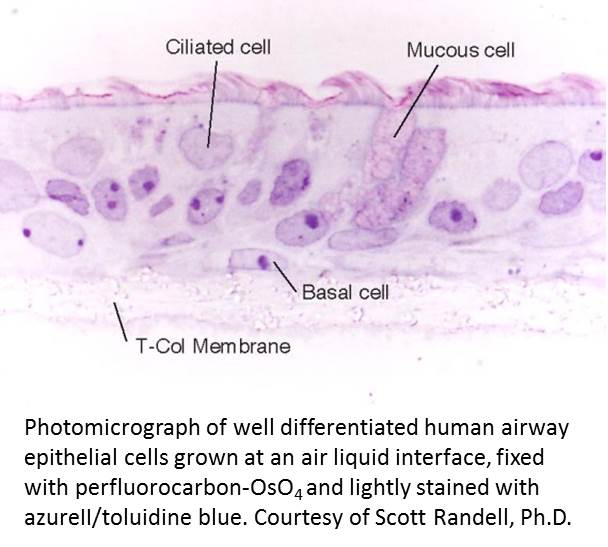 will chronically expose human bronchial and nasal cultures to new and emerging tobacco products (e.g., little cigars and hookah) prior to dissemination to project leaders. Specifically, this core will develop smoke exposure protocols by subjecting representative numbers of cultures to time course and dose response smoke exposure with Kentucky reference cigarettes. The core will then assay acute and chronic LDH release as a primary measure of cell cytotoxicity. Experiments will generally be restricted to those conditions with acute and chronic exposure cell survival >90%. Histology and or confocal analysis will be performed on select cultures after acute and chronic Kentucky reference cigarette smoke exposure. Based on this central data, the core will perform smoke exposures with alternative tobacco
will chronically expose human bronchial and nasal cultures to new and emerging tobacco products (e.g., little cigars and hookah) prior to dissemination to project leaders. Specifically, this core will develop smoke exposure protocols by subjecting representative numbers of cultures to time course and dose response smoke exposure with Kentucky reference cigarettes. The core will then assay acute and chronic LDH release as a primary measure of cell cytotoxicity. Experiments will generally be restricted to those conditions with acute and chronic exposure cell survival >90%. Histology and or confocal analysis will be performed on select cultures after acute and chronic Kentucky reference cigarette smoke exposure. Based on this central data, the core will perform smoke exposures with alternative tobacco 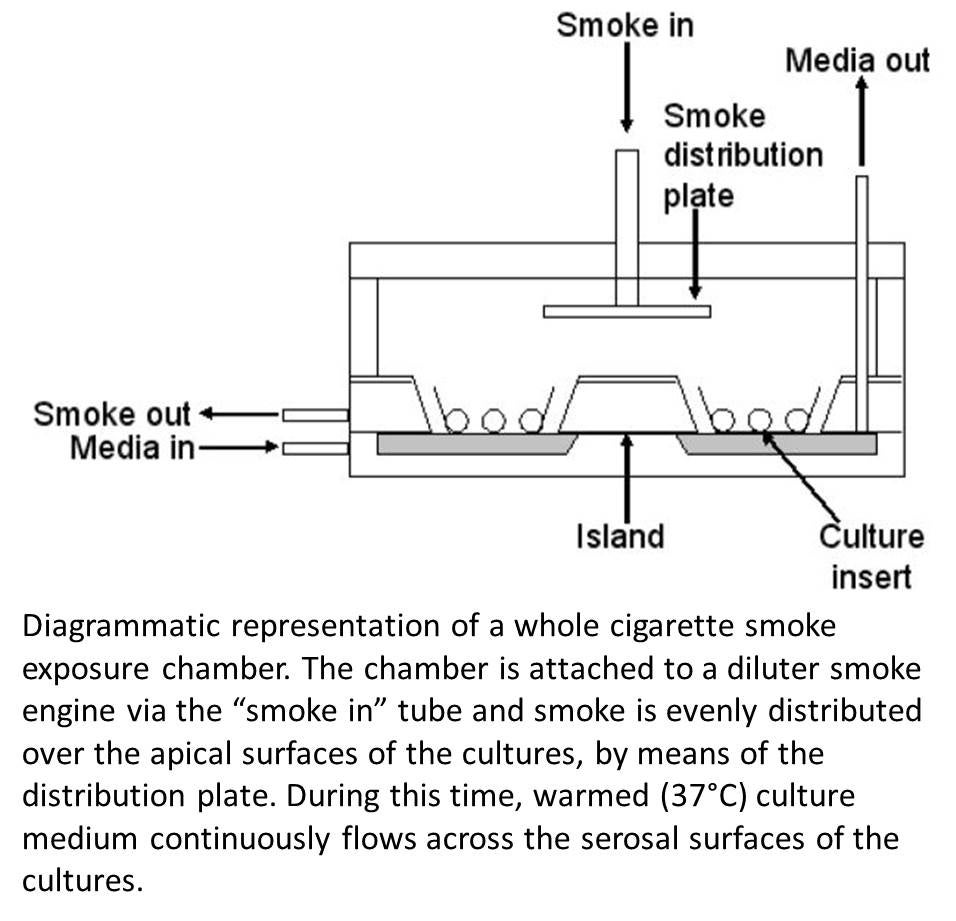 products in a mutually agreed upon schedule as determined in conjunction with project investigators.
products in a mutually agreed upon schedule as determined in conjunction with project investigators.