About Us and Research Services
We offer a variety of in vitro and in vivo research services:
In-Vivo Measurements of Mucociliary and Cough Clearance
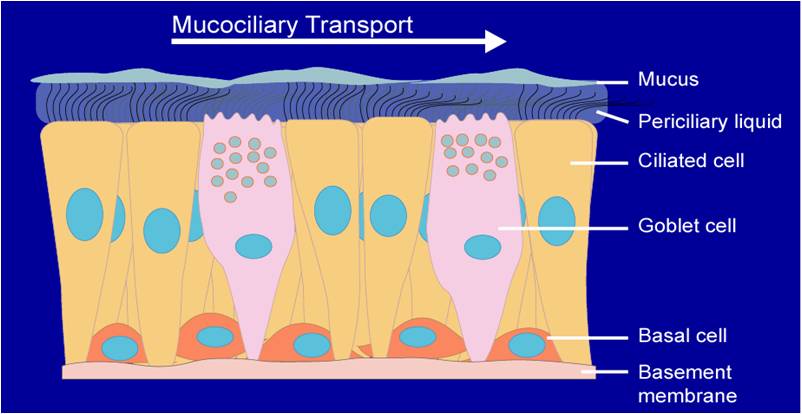
Mucociliary and cough clearance (MCC/CC), is a key part of the innate host defense mechanism important for keeping the airways cleansed of bacteria, viruses and toxic particles. It is impaired in chronic inflammatory airway diseases such as chronic bronchitis (CB), asthma, and cystic fibrosis (CF). Impaired MCC leads to a progressive plugging of the airways with highly viscous, dehydrated mucus that reduces the capacity for ventilation and gas exchange in the lung. In late stage disease, such plugs may only be cleared by cough clearance (CC), which may also become progressively impaired, severely limiting host defense against acute bacterial and viral challenge.
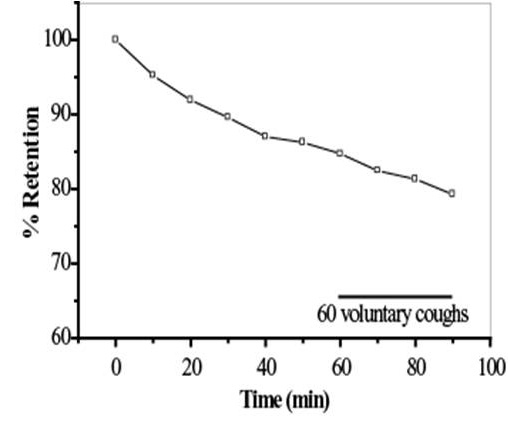
The utility of MCC/CC as a biomarker for disease progression and therapeutic intervention is gaining increased recognition as a valuable tool in the clinical research community (Donaldson et al 2007, Bennett et al 2010 downloads). To measure in-vivo MCC/CC we use inhaled, radiolabeled (Tc99m) sulfur colloid particles that upon deposition in the lung can be followed by gamma camera to determine their rate of egress from the lung. After controlled inhalation of these markers, retention of activity in the lung (as a percent or fraction of initial deposition) is monitored as a function of time over a period of up to 24 hours to determine clearance rates. We can also measure CC by incorporating a fixed number of controlled, voluntary coughs during the measures of radiolabeled particle clearance.
 |
 |
|
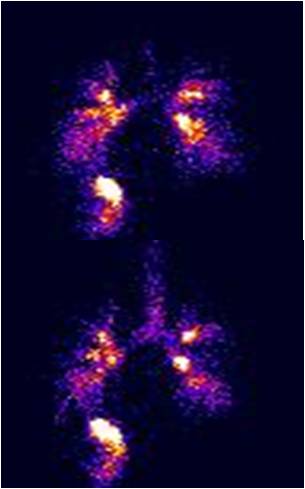
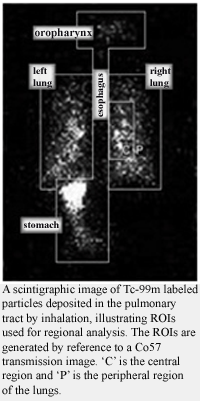
Whole lung clearance is strongly dependent on the site of particle deposition within the lung. To characterize regional deposition in the lung a transmission scan (solid sheet containing Co57 placed in front of the body) of the subject’s lungs is obtained on each subject to outline the area of the whole lung. Only the right lung is generally used to analyze both regional deposition and MCC because of the effects of stomach activity. Central (C) vs. peripheral (P) deposition is determined by regions of interest (ROI) analysis and normalized to C/P for the transmission scan. The whole lung ROI of the right lung is used to determine retention, as a fraction of the initial counts in the right lung, over the clearance period.
 Pulmonary deposition of inhaled aerosol therapies
Pulmonary deposition of inhaled aerosol therapies
We can provide scintigraphic data acquisition and analysis to determine the deposition efficiency in the lung for various aerosol delivery device/drug combinations. For liquid formulations, Tc99m-DTPA or Tc99m-sulfur colloid can be mixed with the drug in solution, while dry powder can be labeled with Tc99m by adsorption onto the powder following the Tc99m extraction from solution by methyl ethyl ketone (MEK). The co-mixtures of radiolabel and drug may then be placed in the device for delivery to the lungs of healthy subjects or patients followed immediately by a posterior gamma scan of the thorax and head/nasal regions. Corrections for differences in attenuation for the various regions (head, lungs, and stomach) may be applied to obtain the relative amounts of activity in each region and for comparison to the initial activity loaded in each device. Aerosol distribution in the lung can also be assessed by C/P ratio and by heterogeneity indices of regional deposition.
To validate our labeling techniques, the particle size output of each device with the co-mixture of drug and radiolabel is analyzed by cascade impaction to ensure that the distribution of the Tc99m is in the same proportion as the drug (see Bench studies below).
Bench studies for measuring output/particle size of aerosol devices
The dose of an aerosol medication delivered to a patient depends on the efficiency of the aerosol delivery system. We use radiolabel techniques to assess delivery and droplet sizes associated with a variety of aerosol delivery devices; nebulizers, dry powder inhalers (DPI), and metered dose inhalers (MDI). In most cases it is very difficult to directly label the drug of interest with a suitable radionuclide. Consequently we use sodium pertechnetate (99mTc-pertechnetate) co-mixed with the drug as an indirect marker to estimate drug delivery and aerosol size distribution. By first showing that the radiolabel co-locates with the nebulized drug, the label can then be used to provide accurate assessments of delivered dose under a variety of conditions.
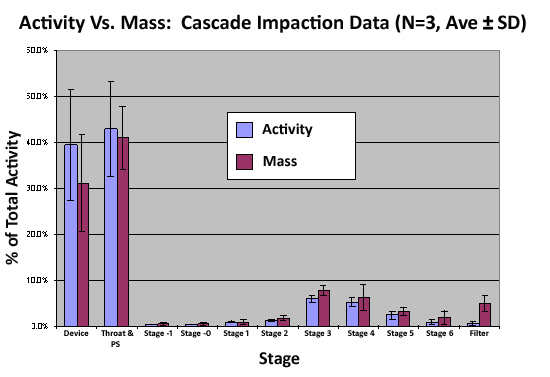
To assess co-localization and sizing of particles the drug-Tc99m aerosol is sampled with a cascade impactor while the particles are being generated from the device under constant flow conditions. The particles are captured on pre-weighed glass fiber cascade impactor stages, dried, and each stage of the impactor counted for radioactivity and weighed. Radioactivity in microcuries(µCi) and drug in nanograms (ng) for each stage of the impactor are plotted and compared for linearity. In order to pass this validation test, quantities of drug and radioactivity on each stage should be equivalent, within the limits of error of the technique, <5%.
 |
 |
 |