Factor XIII
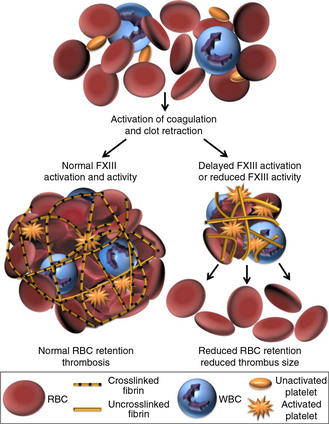
Factor XIII (FXIII) is a protein composed of two catalytic A subunits and two carrier B subunits that circulates in plasma bound to fibrinogen. When activated, the B subunits dissociate, and activated FXIII (FXIIIa) crosslinks glutamine and lysine residues in α- and γ-chains of fibrin monomers, increasing mechanical stability of the clot. Additionally, FXIIIa crosslinks antifibrinolytic plasma proteins to the clot, increasing biochemical stability. We have discovered that FXIII mediates red blood cell retention in clots through fibrin α-chain crosslinking, which in turn mediates clot mass. Genetic or pharmacologic reduction of FXIII leads to the production of smaller thrombi. We have further shown that FXIII zymogen binds fibrinogen in circulation via residues on the fibrinogen γ-chain. Consequently, FXIII could be a novel therapeutic target for preventing venous thrombosis. We are interested in discerning how FXIII mediates venous thrombosis, as well as evaluating potential anti-thrombotic drugs that target FXIII.