Fibrinogen
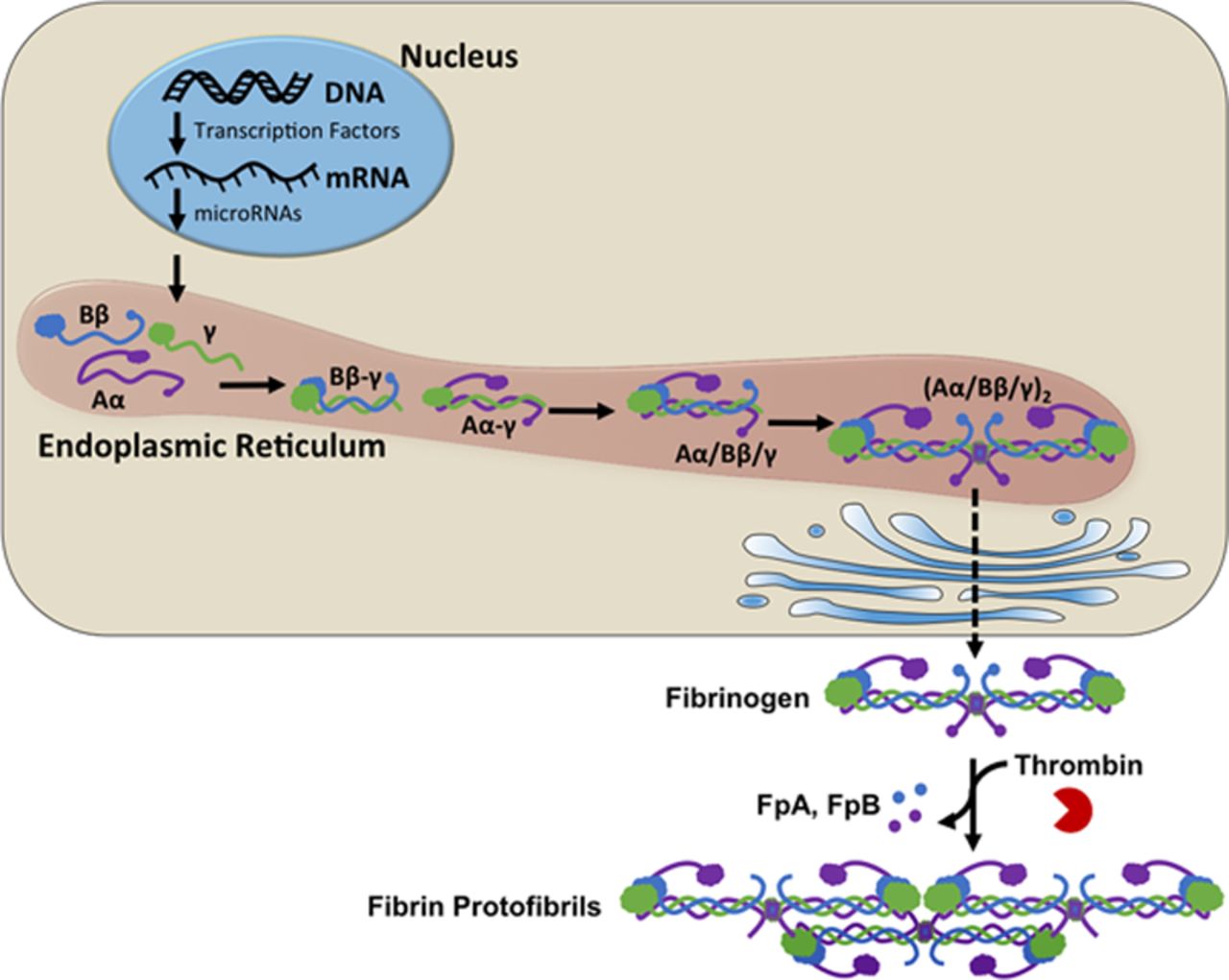
Fibrinogen is a protein composed of 2 Aα-chains, 2 Bβ-chains, and 2 γ-chains that circulates in plasma. During clotting, fibrinogen is cleaved to fibrin by the enzyme thrombin. Fibrin monomers then polymerize into fibers, forming a network that reinforces the clot. Fibrin(ogen) plays a critical role in determining clot formation, structure, and stability, and is thus central to hemostasis and thrombosis. Elevated levels of fibrinogen in the blood lead to increased risk of venous thrombosis. Additionally, fibrin network structure affects risk of venous thrombosis. Multiple factors have been shown to modulate fibrin network structure; the effects of thrombin concentration have been especially well studied. In the presence of high thrombin concentrations, fibrin networks are dense and resistant to fibrinolysis, and in the presence of low thrombin concentrations, fibrin networks are less dense and are susceptible to fibrinolysis. We are interested in elucidating mechanisms leading to altered fibrin structure, as well as investigating the effects of altered fibrin structure on hemostasis and thrombosis.