CASP’s Bill Wilson, PharmD, Clinical Pharmacy Specialist, Infectious Diseases & Antimicrobial Stewardship in the UNC Children’s Hospital, was among the authors on a new article published today in Journal of Oncology Pharmacy Practice.
Recent trials have shown early de-escalation of empiric antimicrobial therapy (EAT) in febrile neutropenia has led to less adverse effects with no difference in patient mortality. In 2019, UNC Hospitals adjusted internal guidelines to de-escalate EAT after seven days of intravenous anti-pseudomonal therapy in patients with signs of clinical recovery from febrile neutropenia and no evidence of infection.
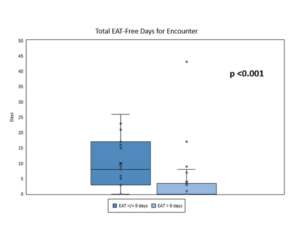 Eligible patients were adults with acute myeloid leukemia (AML) or acute lymphoblastic leukemia (ALL) who received induction chemotherapy and developed febrile neutropenia without documented infection. Patients were separated based on EAT duration: ≤ 9 days and > 9 days. Of the 44 patient encounters reviewed, the authors found that the EAT ≤ 9 days group had 7 more EAT-free days compared to the EAT > 9 days group (p < 0.001). No between-group differences were identified in terms of fever after EAT discontinuation (p = 0.335), ICU admission (p = 0.498), or CDI (p = 0.498). The EAT > 9 days group experienced longer initial fever (p < 0.001) and received addition of resistant Gram-positive coverage (p = 0.014). More patients receiving EAT > 9 days had a diagnosis of AML (p = 0.001).
Eligible patients were adults with acute myeloid leukemia (AML) or acute lymphoblastic leukemia (ALL) who received induction chemotherapy and developed febrile neutropenia without documented infection. Patients were separated based on EAT duration: ≤ 9 days and > 9 days. Of the 44 patient encounters reviewed, the authors found that the EAT ≤ 9 days group had 7 more EAT-free days compared to the EAT > 9 days group (p < 0.001). No between-group differences were identified in terms of fever after EAT discontinuation (p = 0.335), ICU admission (p = 0.498), or CDI (p = 0.498). The EAT > 9 days group experienced longer initial fever (p < 0.001) and received addition of resistant Gram-positive coverage (p = 0.014). More patients receiving EAT > 9 days had a diagnosis of AML (p = 0.001).
The authors concluded that shorter EAT duration did not lead to worse outcomes in patients with AML or ALL who received induction chemotherapy and developed febrile neutropenia without a documented infection source. Read more.