Specialty Areas:
Airway Mucin and Mucus Biochemistry and Proteomics
Research Focus:
One of the main focuses of my work is the characterization of the large mucin gene products (Mr 2-3 million) and the complexes they make (Mr 10-100 million) essential for the formation of the mucus gels vital for epithelial protection and function. My current work is focused around the human lung, where there are many hypersecretory human diseases, including asthma, cystic fibrosis, and chronic bronchitis, in which these glycoconjugates are centrally implicated. Basic understanding of the qualitative and quantitative changes of mucin macromolecules in lung health and diseases is our main task.
Current Projects:
Mucins and Mucin Interactome in the Airway
Mucus is a strong polymer network. The contributions of mucins and other globular proteins, however, have not yet been fully elucidated. The gel-forming mucins, like MUC5AC and MUC5B in particular, are the major constituents of the secreted airway mucus gel. A previously held belief is that they contribute greatly to the biophysical properties of mucus that permit a smooth gel flow on the healthy epithelium in a well-hydrated environment. Despite substantial research on the subject, it is still not clear whether mucin polymers are the key regulators of the biophysical properties of mucus gels. In an effort to address the normal composition of airway mucus, we observed that at least 30% of the proteins in HBE secretion and induced sputum are formed by distinct protein complexes centered around mucins. We propose that these complexes constitute a discrete secretory entity we call the “mucin interactome”. The basis of these structures and their function(s), e.g., and their contribution to the biophysical and biological functions of airway mucus, remain a mystery and are the focus of this project. We hypothesized that mucin-protein interactions dynamically control the critical innate immune functions of the airway mucosal barrier, including its physical (rheological), and biological (antimicrobial, antioxidant) functions. Therefore, as a first step toward testing this hypothesis, HBE cells and a broad range of biochemical, biophysical and proteomic approaches were employed. The aim of this study was to rigorously characterize the airway mucin interactome and study its effects on the basic rheological functions of airway mucus, including hydration, viscosity, and elasticity that provide optimal innate airway defense.
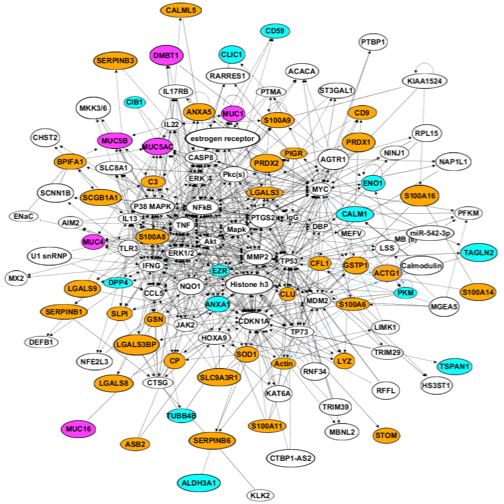
Fig. 1. Cystic Fibrosis in the lung is characterized by the failure of mucus clearance. Clearly mucus plays an important role in all manifestations of CF. There are some two hundred’ mainly globular proteins in human sputum, but it is the gel-forming mucins MUC5AC and MUC5B that we believe create the structural scaffold. The mechanisms underpinning the packaging of the large gel-forming mucins in granules and their subsequent expansion to mature the mucus are very poorly understood. This work is investigating the hypothesis that in the absence of CFTR, mucin expansion/mucus maturation is impaired due to different post-secretory environment in CF mucus. In testing this hypothesis, using the resolving power of the electron microscope in addition to a broad range of biochemical and biophysical techniques, we are testing: 1) To determine the factors effecting the normal maturation of the MUC5B just after secretion; 2) To test the hypothesis that the MUC5B mucin as born into the CF environment is prejudiced in its development and subsequent maturation. Funding: CF foundation (KESIME10I0).
The Effect of Cystic Fibrosis Post-Secretory Settings on Mucin Expansion and Maturation
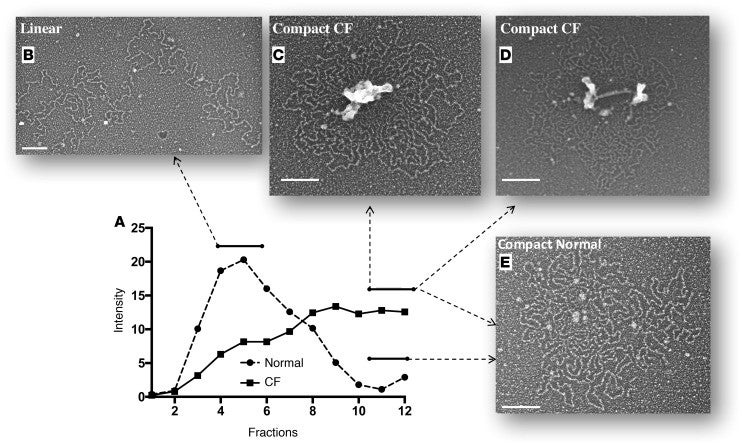
Fig. 2. Transmission electron micrographs of mucin conformation reveal a defective MUC5B unpacking process in CF. Distribution of the molecular forms of salivary MUC5B from a non-CF and CF subject (A) and representative EM images from the top (B) and the bottom (C, D, E) regions. Electron microscopy showed that the compact or semiexpanding granular mucin form of MUC5B is the dominant form found at the bottom of the gradient, and it typically appears as small numbers of glycosylated mucin subunit chains connected around their N- and C-terminal protein nodes in spherical core (10–30 nm) structures (E). In CF samples, however, an analysis of the same fractions indicated that (C and D) the spherical protein nodes/cores are up to 10 times larger (100–300 nm) than (E) their normal equivalents. Scale bars: 100 nm.
Mucin Concentrations and Pathogenesis of Chronic Lung Diseases
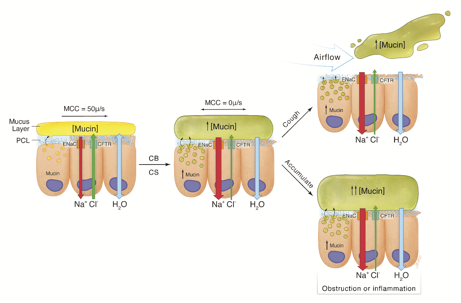
Fig. 3. Relationships of mucin concentrations in sputum with phlegm production and consistency and mucus plug formation. A model representing the relationship from health to cigarette smoke (CS)-induced CB. In health, the balance of active ion absorption (Na+) vs secretion (Cl-), passive osmotically enhanced water transport, and mucin secretion generates a mucus layer with secreted mucin concentrations lower than the tethered mucins and other glycoconjugates in the periciliary layer (PCL). The result is a well hydrated PCL and efficient mucociliary transport. With CB disease/CS exposure, an imbalance of ion transport coupled to mucin hypersecretion increases mucis layer. These events produce adhesion of hyperconcentrated mucus to airway surfaces and. Mucus that cannot be expelled by cough continues to accumulate, concentrates, and ultimately becomes the basis for airflow obstruction and the nidus for infection.
Role of Human Airway Epithelium-Derived Exosome-Like Vesicles in Innate Defense of the Lung
Exosomes are small, 50-150 nm, organelles secreted by different cell types including epithelial, haematopoietic and some tumour cells. The molecular organisation of these structures depends mainly on the cellular source from which they are derived. The detailed roles of the exosomes remain largely unexplained. We have demonstrated their presence in lung secretions and propose that they play important roles in innate lung defence, such as binding to and neutralizing the viruses. The objective of this work is to elucidate the organization of the human tracheobronchial epithelial (HTBE) exosomes and identify their cellular origin, ultimately we wish to understand their roles in airway host defense. Our preliminary data from HTBE cell culture indicate that exosomes are highly organized structures arising from different cell types to be found in the culture and that they interact with human influenza virus. Many of them have membrane tethered mucin coats that control their recognition properties. Funding: American Lung Association (RG-167538-N).
Selected Bibliography:
- Kesimer M, Radicioni G, Ford AA, Ceppe A, Alexis NE, Barr RG, Bleecker ER, Christenson SA, Cooper CB, Han MK, Hansel NN, Hastie AT, Hoffman EA, Kanner RE, Martinez FJ, Paine R, Woodruff PG, Boucher RC. A Quantitative Lung Mucin Score to Identify Chronic Bronchitis. NEJM Evid. 2026 Feb;5(2):EVIDoa2500051. doi: 10.1056/EVIDoa2500051. PMID: 41590989. PMCID: PMC13055890.
- Sun L, Walls SA, Dang H, Quinney NL, Sears PR, Sadritabrizi T, Hasegawa K, Okuda K, Asakura T, Chang X, Zheng M, Mikami Y, Dizmond FU, Danilova D, Zhou L, Deshmukh A, Cholon DM, Radicioni G, Rogers TD, Kissner WJ, Markovetz MR, Guhr Lee TN, Gutay MI, Esther CR Jr, Chua M, Grubb BR, Ehre C, Kesimer M, Hill DB, Ostrowski LE, Button B, Gentzsch M, Robinson C, Olivier KN, Freeman AF, Randell SH, Vladar E, O’Neal WK, Boucher RC Jr, Chen G. STAT3-dependent Regulation of CFTR and Ciliogenesis Is Essential for Mucociliary Clearance and Innate Airway Defense in Hyper-IgE Syndrome. Am J Respir Crit Care Med. 2025 Oct;211(10):1951-1969. doi: 10.1164/rccm.202407-1415OC. PMID: 40315437. PMCID: PMC12555045.
- Ozkan E, Livengood SS, Ford AA, Macdonald JK, Samir S, Klevans IW, Kesimer M. Analytical validation of total mucin concentration assay using SEC MALLS dRI for diagnosing and monitoring mucoobstructive lung diseases. Sci Rep. 2025 Apr 29;15(1):15024. doi: 10.1038/s41598-025-97808-4. PMID: 40301448; PMCID: PMC12041582.
- Curtis JL, Bateman LA, Murray S, Couper DJ, Labaki WW, Freeman CM, Arnold KB, Christenson SA, Alexis NE, Kesimer M, Boucher RC, Kaner RJ, Barjaktarevic I, Cooper CB, Hoffman EA, Barr RG, Bleecker ER, Bowler RP, Comellas A, Dransfield MT, Freedman MB, Hansel NN, Krishnan JA, Marchetti N, Meyers DA, Ohar J, O’Neal WK, Ortega VE, Paine Iii R, Peters SP, Smith BM, Wedzicha JA, Wells JM, Woodruff PG, Han MK, Martinez FJ. Design of the SPIROMICS Study of Early COPD Progression: SOURCE Study. Chronic Obstr Pulm Dis. 2024 Sep 27;11(5):444-459. doi: 10.15326/jcopdf.2023.0490. PMID: 39159077. PMCID: PMC11548966.
- Opron K, Begley LA, Erb-Downward JR, Li G, Alexis NE, Barjaktarevic I, Barr RG, Bleecker ER, Boucher R, Bowler RP, Christenson SA, Comellas AP, Criner G, Cooper CB, Couper D, Galban CJ, Han MK, Hastie A, Hatt C, Hoffman EA, Kaner RJ, Kesimer M, Krishnan JA, LaFon DC, Martinez FJ, Ortega VE, Peters SP, Paine Iii R, Putcha N, Woodruff PG, Huffnagle GB, Kozik AJ, Curtis JL, Huang YJ; SPIROMICS Investigators. Loss of Airway Phylogenetic Diversity Is Associated with Clinical and Pathobiological Markers of Disease Development in COPD. Am J Respir Crit Care Med. 2024 Jul 15;210(2):186-200. doi: 10.1164/rccm.202303-0489OC. PMID: 38261629. PMCID: PMC11273318.
- Greenwald MA, Meinig SL, Plott LM, Roca C, Higgs MG, Vitko NP, Markovetz MR, Rouillard KR, Carpenter J, Kesimer M, Hill DB, Schisler JC, Wolfgang MC. Mucus polymer concentration and in vivo adaptation converge to define the antibiotic response of Pseudomonas aeruginosa during chronic lung infection. mBio. 2024 Jun 12;15(6):e0345123. doi: 10.1128/mbio.03451-23. PMID: 38651896. PMCID: PMC11237767.
- Asakura T, Okuda K, Chen G, Dang H, Kato T, Mikami Y, Schworer SA, Gilmore RC, Radicioni G, Hawkins P, Barbosa Cardenas SM, Saito M, Cawley AM, De la Cruz G, Chua M, Alexis NE, Masugi Y, Noone PG, Ribeiro CMP, Kesimer M, Olivier KN, Hasegawa N, Randell SH, O’Neal WK, Boucher RC. Proximal and Distal Bronchioles Contribute to the Pathogenesis of Non-Cystic Fibrosis Bronchiectasis (NCFB). Am J Respir Crit Care Med. 2024 Feb 15;209(4):374-389. doi: 10.1164/rccm.202306-1093OC. PMID: 38016030. PMCID: PMC10878387.
- Carpenter J, Kesimer M. Imaging of Mucin Networks with Atomic Force Microscopy. Methods Mol Biol. 2024;2763:361-371. doi: 10.1007/978-1-0716-3670-1_31. PMID: 38347426. PMCID: PMC11127370.
- Carpenter J, Kesimer M. Isolation of Membrane Bound Mucins from Human Bronchial Epithelial Cells. Methods Mol Biol. 2024;2763:51-59. doi: 10.1007/978-1-0716-3670-1_4. PMID: 38347399. PMCID: PMC11149713.
- Radicioni G, Kesimer M. Quantitation of MUC5AC and MUC5B by Stable Isotope Labeling Mass Spectrometry. Methods Mol Biol. 2024;2763:125-136. doi: 10.1007/978-1-0716-3670-1_11. PMID: 38347406.
- Lee RE, Reidel B, Nelson MR, Macdonald JK, Kesimer M, Randell SH. Air-Liquid interface cultures to model drug delivery through the mucociliary epithelial barrier. Adv Drug Deliv Rev. 2023 Jul;198:114866. doi: 10.1016/j.addr.2023.114866. PMID: 37196698. PMCID: PMC10336980.
- Van Buren E, Radicioni G, Lester S, O’Neal WK, Dang H, Kasela S, Garudadri S, Curtis JL, Han MK, Krishnan JA, Wan ES, Silverman EK, Hastie A, Ortega VE, Lappalainen T, Nawijn MC, Berge MVD, Christenson SA, Li Y, Cho MH, Kesimer M, Kelada SNP. Genetic regulators of sputum mucin concentration and their associations with COPD phenotypes. PLoS Genet. 2023 Jun 23;19(6):e1010445. doi: 10.1371/journal.pgen.1010445. PMID: 37352370; PMCID: PMC10325042.
- Reidel B, Abdelwahab S, Wrennall JA, Clapp PW, Beers JL, Jackson KD, Tarran R, Kesimer M. Vaping additives cannabinoid oil and vitamin E acetate adhere to and damage the human airway epithelium. J Appl Toxicol. 2023 May;43(5):680-693. doi: 10.1002/jat.4415. PMID: 36372912. PMCID: PMC10101868.
- Kesimer M. Mucins MUC5AC and MUC5B in the airways: MUCing around together. Am J Respir Crit Care Med. 2022 Nov 1;206(9):1055-1057. doi: 10.1164/rccm.202208-1459ED. PMID: 35938865. PMCID: PMC9704829.
- Iverson E, Griswold K, Song D, Gagliardi TB, Hamidzadeh K, Kesimer M, Sinha S, Perry M, Duncan GA, Scull MA. Membrane-Tethered Mucin 1 Is Stimulated by Interferon and Virus Infection in Multiple Cell Types and Inhibits Influenza A Virus Infection in Human Airway Epithelium. mBio. 2022 Aug 30;13(4):e0105522. doi: 10.1128/mbio.01055-22. PMID: 35699372. PMCID: PMC9426523.
- Batson B, Zorn B, Radicioni G, Livengood S, Kumagai T, Dang H, Ceppe A, Clapp P, Tunney M, Elborn S, McElvaney G, Muhlebach M, Boucher RC, Tiemeyer M, Wolfgang M, Kesimer M. Cystic Fibrosis Airway Mucus Hyperconcentration Produces a Vicious Cycle of Mucin, Pathogen, and Inflammatory Interactions that Promote Disease Persistence. Am J Respir Cell Mol Biol. 2022 Aug;67(2):253-265. doi: 10.1165/rcmb.2021-0359OC. PMID: 35486871. PMCID: PMC9348562.
- Esther CR Jr, O’Neal WK, Anderson WH, Kesimer M, Ceppe A, Doerschuk CM, Alexis NE, Hastie AT, Barr RG, Bowler RP, Wells JM, Oelsner EC, Comellas AP, Tesfaigzi Y, Kim V, Paulin LM, Cooper CB, Han MK, Huang YJ, Labaki WW, Curtis JL, Boucher RC; SPIROMICS. Identification of Sputum Biomarkers Predictive of Pulmonary Exacerbations in Chronic Obstructive Pulmonary Disease. Chest. 2022 May;161(5):1239-1249. doi: 10.1016/j.chest.2021.10.049. PMID: 34801592. PMCID: PMC9131049.
- Singanayagam A, Footitt J, Marczynski M, Radicioni G, Cross MT, Finney LJ, Trujillo-Torralbo MB, Calderazzo MA, Zhu J, Aniscenko J, Clarke TB, Molyneaux PL, Bartlett NW, Moffatt MF, Cookson WO, Wedzicha JA, Evans CM, Boucher RC, Kesimer M, Lieleg O, Mallia P, Johnston SL. Airway mucins promote immunopathology in virus-exacerbated chronic obstructive pulmonary disease. J Clin Invest. 2022 Apr 15;132(8):e120901. doi: 10.1172/JCI120901. PMID: 35239513. PMCID: PMC9012283.
- Costain G, Liu Z, Mennella V, Radicioni G, Goczi AN, Albulescu A, Walker S, Ngan B, Manson D, Vali R, Khan M, Palaniyar N, Hill DB, Hall DA, Marshall CR, Knowles M, Zariwala MA, Kesimer M, Dell SD. Hereditary Mucin Deficiency Caused by Biallelic Loss-of-Function of MUC5B. Am J Respir Crit Care Med. 2022 Apr 1;205(7):761-768. doi: 10.1164/rccm.202106-1456OC. PMID: 35023825. PMCID: PMC9836224.
- Kato T, Radicioni G, Papanikolas MJ, Stoychev GV, Markovetz MR, Aoki K, Porterfield M, Okuda K, Barbosa Cardenas SM, Gilmore RC, Morrison CB, Ehre C, Burns KA, White KK, Brennan TA, Goodell HP, Thacker H, Loznev HT, Forsberg LJ, Nagase T, Rubinstein M, Randell SH, Tiemeyer M, Hill DB, Kesimer M, O’Neal WK, Ballard ST, Freeman R, Button B, Boucher RC. Mucus concentration-dependent biophysical abnormalities unify submucosal gland and superficial airway dysfunction in cystic fibrosis. Sci Adv. 2022 Apr;8(13):eabm9718. doi: 10.1126/sciadv.abm9718. PMID: 35363522.
- Morrison CB, Shaffer KM, Araba KC, Markovetz MR, Wykoff JA, Quinney NL, Hao S, Delion MF, Flen AL, Morton LC, Liao J, Hill DB, Drumm ML, O’Neal WK, Kesimer M, Gentzsch M, Ehre C. Treatment of cystic fibrosis airway cells with CFTR modulators reverses aberrant mucus properties via hydration. Eur Respir J. 2022 Feb 3;59(2):2100185. doi: 10.1183/13993003.00185-2021. PMID: 34172469. PMCID: PMC8859811.
- Choudhary I, Vo T, Paudel K, Wen X, Gupta R, Kesimer M, Patial S, Saini Y. Vesicular and extravesicular protein analyses from the airspaces of ozone-exposed mice revealed signatures associated with mucoinflammatory lung disease. Sci Rep. 2021 Dec 1;11(1):23203. doi: 10.1038/s41598-021-02256-5. PMID: 34853335; PMCID: PMC8636509.
- Woodall M, Reidel B, Kesimer M, Tarran R, Baines DL. Culture with apically applied healthy or disease sputum alters the airway surface liquid proteome and ion transport across human bronchial epithelial cells. Am J Physiol Cell Physiol. 2021 Dec 1;321(6):C954-C963. doi: 10.1152/ajpcell.00234.2021. PMID: 34613844. PMCID: PMC8714986.
- Radicioni G, Ceppe A, Ford AA, Alexis NE, Barr RG, Bleecker ER, Christenson SA, Cooper CB, Han MK, Hansel NN, Hastie AT, Hoffman EA, Kanner RE, Martinez FJ, Ozkan E, Paine R 3rd, Woodruff PG, O’Neal WK, Boucher RC, Kesimer M. Airway mucin MUC5AC and MUC5B concentrations and the initiation and progression of chronic obstructive pulmonary disease: an analysis of the SPIROMICS cohort. Lancet Respir Med. 2021 Nov;9(11):1241-1254. doi: 10.1016/S2213-2600(21)00079-5. PMID: 34058148. PMCID: PMC8570975.
- Carpenter J, Wang Y, Gupta R, Li Y, Haridass P, Subramani DB, Reidel B, Morton L, Ridley C, O’Neal WK, Buisine MP, Ehre C, Thornton DJ, Kesimer M. Assembly and organization of the N-terminal region of mucin MUC5AC: Indications for structural and functional distinction from MUC5B. Proc Natl Acad Sci U S A. 2021 Sep 28;118(39):e2104490118. doi: 10.1073/pnas.2104490118. PMID: 34548396; PMCID: PMC8488587.
- Carpenter J, Kesimer M. Membrane-bound mucins of the airway mucosal surfaces are densely decorated with keratan sulfate: revisiting their role in the Lung’s innate defense. Glycobiology. 2021 May 3;31(4):436-443. PMID: 33083824; PMCID: PMC8091473.
- Abdelwahab SH, Reidel B, Martin JR, Ghosh A, Keating JE, Haridass P, Carpenter J, Glish GL, Tarran R, Doerschuk CM, Kesimer M. Cigarillos Compromise the Mucosal Barrier and Protein Expression in Airway Epithelia. Am J Respir Cell Mol Biol. 2020 Dec;63(6):767-779. PMID: 32877614; PMCID: PMC7790145.
- Kim V, Jeong S, Zhao H, Kesimer M, Boucher RC, Wells JM, Christenson SA, Han MK, Dransfield M, Paine R 3rd, Cooper CB, Barjaktarevic I, Bowler R, Curtis JL, Kaner RJ, O’Beirne SL, O’Neal WK, Rennard SI, Martinez FJ, Woodruff PG. Current smoking with or without chronic bronchitis is independently associated with goblet cell hyperplasia in healthy smokers and COPD subjects. Sci Rep. 2020 Nov 18;10(1):20133. PMID: 33208859; PMCID: PMC7674445.
- McQueen BE, Kiatthanapaiboon A, Fulcher ML, Lam M, Patton K, Powell E, Kollipara A, Madden V, Suchland RJ, Wyrick P, O’Connell CM, Reidel B, Kesimer M, Randell SH, Darville T, Nagarajan UM. Human Fallopian Tube Epithelial Cell Culture Model To Study Host Responses to Chlamydia trachomatis Infection. Infect Immun. 2020 Aug 19;88(9):e00105-20. PMID: 32601108; PMCID: PMC7440757.
- Ramsey KA, Chen ACH, Radicioni G, Lourie R, Martin M, Broomfield A, Sheng YH, Hasnain SZ, Radford-Smith G, Simms LA, Burr L, Thornton DJ, Bowler SD, Livengood S, Ceppe A, Knowles MR, Noone PG, Donaldson SH, Hill DB, Ehre C, Button B, Alexis NE, Kesimer M, Boucher RC, McGuckin MA. Airway Mucus Hyperconcentration in Non-Cystic Fibrosis Bronchiectasis. Am J Respir Crit Care Med. 2020 Mar 15;201(6):661-670. PMID: 31765597; PMCID: PMC7068838.
- Ridley C, Lockhart-Cairns MP, Collins RF, Jowitt TA, Subramani DB, Kesimer M, Baldock C, Thornton DJ. The C-terminal dimerization domain of the respiratory mucin MUC5B functions in mucin stability and intracellular packaging before secretion. J Biol Chem. 2019 Nov 8;294(45):17105-17116. PMID: 31570524; PMCID: PMC6851316.
- Kesimer M. Another Warning Sign: High Nicotine Content in E-Cigarettes Disrupts Mucociliary Clearance, the Essential Defense Mechanism of the Lung. Am J Respir Crit Care Med. 2019 Nov 1;200(9):1082-1084. PMID: 31199664; PMCID: PMC6888647.
- Chen G, Sun L, Kato T, Okuda K, Martino MB, Abzhanova A, Lin JM, Gilmore RC, Batson BD, Volmer AS, Dang H, Deng Y, Randell SH, Button B, Livraghi-Butrico A, Kesimer M, Ribeiro CMP, O’Neal WK, Boucher RC. IL-1β dominates the promucin secretory cytokine profile in cystic fibrosis. J Clin Invest. 2019 Oct 1;129(10):4433-4450. PMID: 31524632; PMCID: PMC6763234.
- Markovetz MR, Subramani DB, Kissner WJ, Morrison CB, Garbarine IC, Ghio A, Ramsey KA, Arora H, Kumar PA, Nix DB, Kumagai T, Krunkosky TM, Krause DC, Radicioni G, Alexis NE, Kesimer M, Tiemeyer M, Boucher RC, Ehre C, Hill DB. Endotracheal Tube Mucus as a Source of Airway Mucus for Rheological Study. Am J Physiol Lung Cell Mol Physiol. 2019 Oct 1;317(4):L498-L509. PMID: 31389736; PMCID: PMC6842913.
- Esther CR Jr, Muhlebach MS, Ehre C, Hill DB, Wolfgang MC, Kesimer M, Ramsey KA, Markovetz MR, Garbarine IC, Forest MG, Seim I, Zorn B, Morrison CB, Delion MF, Thelin WR, Villalon D, Sabeter JR, Turkovic L, Ranganathan S, Stick SM, Boucher RC, on behalf of AREST CF. Mucus accumulation in the lungs precedes structural changes and infection in children with cystic fibrosis. Sci Transl Med. 2019 Apr 3;11(486). PMID: 30944166; PMCID: PMC6566903.
- Okuda K, Chen G, Subramani DB, Wolf M, Gilmore RC, Kato T, Radicioni G, Kesimer M, Chua M, Livraghi-Butrico A, Ehre C, Doerschuk CM, Randell SH, Matsui H, Nagase T, O’Neal WK, Boucher RC. Localization of secretory mucins MUC5AC and MUC5B in normal human airways. Am J Respir Crit Care Med. 2019 Mar 15;199(6):715-727. PMID: 30352166; PMCID: PMC6423099.
- Gupta R, Radicioni G, Abdelwahab S, Dang H, Carpenter J, Chua M, Mieczkowski PA, Sheridan J, Randell SH, Kesimer M. Intercellular Communication between Airway Epithelial Cells is Mediated by Exosome-Like Vesicles. Am J Respir Cell Mol Biol. 2019 Feb;60(2):209-220. PMID: 30230353; PMCID: PMC6376407.
- Henderson AG, Anderson WH, Ceppe A, Coakley RD, Button B, Alexis NE, Peden DB, Lazarowski ER, Davis CW, Fuller F, Almond M, Qaqish B, Kesimer M, Boucher RC. Mucus hydration in subjects with stable chronic bronchitis: A comparison of spontaneous and induced sputum. COPD. 2018 Dec;15(6):572-580. PMID: 30712400; PMCID: PMC6428611.
- Kesimer M, Smith BM, Ceppe A, Ford AA, Anderson WH, Barr RG, O’Neal W, Boucher RC; for SPIROMICS Investigative Group. Mucin Concentrations and Peripheral Airways Obstruction in COPD. Am J Respir Crit Care Med. 2018 Dec 1;198(11):1453-1456. PMID: 30130124; PMCID: PMC6290944.
- Martinez FJ, Han ML, Allinson JP, Barr RG, Boucher RC, Calverley PMA, Celli BR, Christenson S, Crystal RG, Fagerås M, Freeman C, Gröenke L, Hoffman EA, Kesimer M, Kostikas K, Paine R III, Rafii S, Rennard SI, Segal LN, Stevenson C, Tal-Singer R, Vestbo J, Woodruff PG, Curtis JL, Wedzicha JA. At the root: Defining and halting progression of early chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018 Jun 15;197(12):1540-1551. PMID: 29406779; PMCID: PMC6006401.
- Moore PJ, Reidel B, Ghosh A, Sesma J, Kesimer M, Tarran R. Cigarette smoke modifies and inactivates SPLUNC1, leading to airway dehydration. FASEB J. 2018 Jun 11:fj201800345R. PMID: 29890087; PMCID: PMC6219833.
- Abdullah LH, Coakley R, Webster MJ, Zhu Y, Tarran R, Radicioni G, Kesimer M, Boucher RC, Davis CW, Ribeiro CMP. Mucin production and hydration responses to mucopurulent materials in normal vs. CF airway epithelia. Am J Respir Crit Care Med. 2018 Feb 15;197(4):481-491. PMID: 29099608; PMCID: PMC5821906.
- Terryah ST, Fellner RC, Ahmad S, Moore PJ, Reidel B, Sesma JI, Kim CS, Garland AL, Scott DW, Sabater JR, Carpenter J, Randell SH, Kesimer M, Abraham WM, Arendshorst WJ, Tarran R. Evaluation of a SPLUNC1-derived peptide for the treatment of cystic fibrosis lung disease. Am J Physiol Lung Cell Mol Physiol. 2018 Jan 1;314(1):L192-L205. PMID: 28982737; PMCID: PMC5866433.
- Kesimer M, Ford AA, Ceppe A, Radicioni G, Cao R, Davis CW, Doerschuk CM, Alexis NE, Anderson WH, Henderson AG, Barr G, Bleecker ER, Christenson SA, Cooper CB, Han MLK, Hansel NN, Hastie AT, Hoffman EA, Kanner RE, Martinez F, Paine R, Woodruff PG, O’Neal WK, Boucher RC. Airway Mucin Concentration as a Marker of Chronic Bronchitis. N Engl J Med. 2017;377:911-22. PMID: 28877023; PMCID: PMC5706541.
- Reidel B, Radicioni G, Clapp PW, Ford AA, Abdelwahab S, Rebuli ME, Haridass P, Alexis NE, Jaspers I, Kesimer M. E-Cigarette Use Causes a Unique Innate Immune Response in the Lung Involving Increased Neutrophilic Activation and Altered Mucin Secretion. Am J Respir Crit Care Med. 2018 Feb 15;197(4):481-491. PMID: 29053025; PMCID: PMC5821909.
- Adams DC, Pahlevaninezhad H, Szabari MV, Cho JL, Hamilos DL, Kesimer M, Boucher RC, Luster AD, Medoff BD, Suter MJ. Automated segmentation and quantification of airway mucus with endobronchial optical coherence tomography. Biomed Opt Express. 2017;8:4729-41. PMID: 29082098; PMCID: PMC5654813.
- Burbank AJ, Duran CG, Pan Y, Burns P, Jones S, Jiang Q, Yang C, Jenkins S, Wells H, Alexis N, Kesimer M, Bennett WD, Zhou H, Peden DB, Hernandez ML. Gamma tocopherol-enriched supplement reduces sputum eosinophilia and endotoxin-induced sputum neutrophilia in volunteers with asthma. J Allergy Clin Immunol. 2018 Apr;141(4):1231-1238.e1. PMID: 28736267; PMCID: PMC5775926.
- Ghosh A, Abdelwahab SH, Reeber SL, Reidel B, Marklew AJ, Garrison AJ, Lee S, Dang H, Herring AH, Glish GL, Kesimer M, Tarran R. Little Cigars are More Toxic than Cigarettes and Uniquely Change the Airway Gene and Protein Expression. Sci Rep. 2017 Apr 27;7:46239. PMID: 28447619; PMCID: PMC5406835.
- Abdullah LH, Evans JR, Wang TT, Ford AA, Makhov AM, Nguyen K, Coakley RD, Griffith JD, Davis CW, Ballard ST, Kesimer M. Defective postsecretory maturation of MUC5B mucin in cystic fibrosis airways. JCI Insight. 2017 Mar 23;2(6):e89752. PMID: 28352653; PMCID: PMC5358479.
- Dickey BF, Fahy JV, Kesimer M, Boucher RC, Evans CM, Thornton D. Measuring Airway Mucin 2 in Patients with Severe Chronic Obstructive Pulmonary Disease with Bacterial Colonization. Ann Am Thorac Soc. 2016;13:2103-4. PMID: 27831803; PMCID: PMC5802559.
- Cho JL, Ling MF, Adams DC, Faustino L, Islam SA, Afshar R, Griffith JW, Harris RS, Ng A, Radicioni G, Ford AA, Han AK, Xavier R, Kwok WW, Boucher R, Moon JJ, Hamilos DL, Kesimer M, Suter MJ, Medoff BD, Luster AD. Allergic asthma is distinguished by sensitivity of allergen-Specific CD4+ T Cells and Airway Structural Cells to Type 2 Inflammation. Sci Transl Med. 2016 Oct 5;8(359):359ra132.2016. PMID: 27708065; PMCID: PMC5399547.
- Woodruff PG, Barr RG, Bleecker E, Christenson SA, Couper D, Curtis JL, Gouskova NA, Hansel NN, Hoffman EA, Kanner RE, Kleerup E, Lazarus SC, Martinez FJ, Paine R 3rd, Rennard S, Tashkin DP, Han MK; SPIROMICS Research Group. Clinical Significance of Symptoms in Smokers with Preserved Pulmonary Function. N Engl J Med. 2016 May 12;374(19):1811-21. PMID: 27168432; PMCID: PMC4968204.
- Radicioni G, Cao R, Carpenter, J. Ford. AA., Wang T, Li L, Kesimer M. Innate immune properties of airway epithelial surfaces are regulated by dynamic interactions between mucins and interacting proteins: the mucin interactome. Mucosal Immunol. 2016, 9, 1442-1454. PMID: 27072609; PMCID: PMC5063652. Featured as cover article of December 2017.
- Kesimer M, Cullen J, Cao R, Radicioni G, Mathews K, Seiler G, Gookin J. Excess secretion of gel-forming mucins and associated innate defense proteins with defective mucin un-packaging underpin gall bladder mucocele formation in dogs. PLoS One. 2015 Sep 28;10(9):e0138988. PMID: 26414376; PMCID: PMC4586375.
- Kesimer M, Gupta R. Physical characterization and profiling of airway epithelial derived exosomes using light scattering. Methods. 2015 Oct 1;87:59-63. PMID: 25823850; PMCID: PMC4584172.
- Anderson WH, Coakley RD, Button B, Henderson AG, Zeman KL, Alexis NE, Peden DB, Lazarowski ER, Davis CW, Bailey S, Fuller F, Almond M, Qaqish B, Bordonali E, Rubinstein M, Bennett WD, Kesimer M, Boucher RC. The Relationship of Mucus Concentration (Hydration) to Mucus Osmotic Pressure and Transport in Chronic Bronchitis. Am J Respir Crit Care Med. 2015 Jul 15;192(2):182-90. PMID: 25909230; PMCID: PMC4532825.
- Henderson AG, Ehre C, Button B, Abdullah LH, Cai LH, Leigh MW, DeMaria GC, Matsui H, Donaldson SH, Davis CW, Sheehan JK, Boucher RC, Kesimer M. Cystic fibrosis airway secretions exhibit mucin hyperconcentration and increased osmotic pressure. J Clin Invest. 2014 Jul;124(7):3047-60. PMID: 24892808; PMCID: PMC4072023.
- Hobbs CA, Blanchard MG, Alijevic O, Tan CD, Kellenberger S, Bencharit S, Cao R, Kesimer M, Walton WG, Henderson AG, Redinbo MR, Stutts MJ, Tarran R. Identification of the SPLUNC1 ENaC-inhibitory domain yields novel strategies to treat sodium hyperabsorption in cystic fibrosis airway epithelial cultures. Am J Physiol Lung Cell Mol Physiol. 2013 Dec;305(12):L990-L1001. Erratum in: Am J Physiol Lung Cell Mol Physiol. 2014 Apr 1;306(7):L708. PMID: 24124190; PMCID: PMC3882538.
- Couper D, LaVange LM, Han M, Barr RG, Bleecker E, Hoffman EA, Kanner R, Kleerup E, Martinez FJ, Woodruff PG, Rennard S; for the SPIROMICS Research Group. Design of the Subpopulations and Intermediate Outcomes in COPD Study (SPIROMICS). Thorax. 2014 May;69(5):491-4. PMID: 24029743; PMCID: PMC3954445.
- Kesimer M, Ehre C, Burns KA, Davis CW, Sheehan JK, Pickles RJ. Molecular Organization of the Mucins and Glycocalyx Underlying Mucus Transport Over Mucosal Surfaces of the Airways. Mucosal Immunol. 2013 Mar;6(2):379-92. PMID: 22929560; PMCID: PMC3637662. Featured as cover article of March 2013.
- Button B, Cai JH, Ehre C, Kesimer M, Hill DB, Sheehan JK, Boucher RC, Rubinstein M. A Periciliary Brush Promotes the Lung Health by Separating the Mucus Layer from Airway Epithelia. Science. 2012 Aug 24;337(6097):937-41. PMID: 22923574; PMCID: PMC3633213. Featured as Cover article.
- Guo X, Kesimer M, Tolun G, Zheng X, Xu Q, Lu J, Sheehan JK, Griffith JD, Li X. The NAD(+)-dependent protein deacetylase activity of SIRT1 is regulated by its oligomeric status. Sci Rep. 2012;2:640. PMID: 22962634; PMCID: PMC3435561.
- Gercel-Taylor C, Atay S, Tullis RH, Kesimer M, Taylor DD. Nanoparticle analysis of circulating cell-derived vesicles in ovarian cancer patients. Anal Biochem. 2012 Sep 1;428(1):44-53. PMID: 22691960.
- Cao R, Wang TT, DeMaria G, Sheehan JK, Kesimer M. Mapping the protein domain structures of the respiratory mucins: a mucin proteome coverage study. J Proteome Res. 2012 Aug 3;11(8):4013-23. PMID: 22663354; PMCID: PMC3412937.
- Kesimer M, Makhov AM, Griffith JD, Verdugo P, Sheehan JK. Unpacking a gel-forming mucin: a view of MUC5B organization after granular release. Am J Physiol Lung Cell Mol Physiol. 2010 Jan;298(1):L15-22. PMID: 19783639; PMCID: PMC2806194. Published as “Editorial Focus”. Commented by Quinton PM. Birth of mucus. Am J Physiol Lung Cell Mol Physiol. 2010 Jan;298(1):L13-4.
