Research Interests
- Signaling networks controlling cell function and disease
Research Synopsis
My laboratory defines the “signal relay” systems initiated by various cellular stimuli including cytokines, growth factors, antigens, and drugs used to treat human disease.The unifying hypothesis directing the work in my laboratory is that the differential expression and spatial organization of signal relay proteins controls the responsiveness of different cell types to specific stimuli. Aberrant spatial and temporal control of signal relay systems is a major contributing factor to human diseases including cancer, cardiovascular disease and auto-immunity. The primary signal relay systems studied in the lab involve the mitogen activated protein kinase (MAPK) pathways and how they regulate gene expression, cell growth, development and apoptosis.
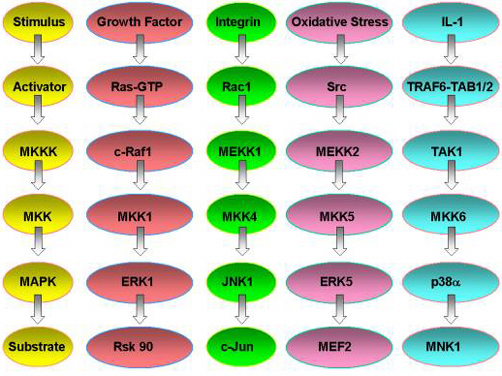
MAP kinase pathways are composed of three kinases: a MAP kinase (MAPK), MAPK kinase (MKK), and a MAPK kinase kinase (MKKK). Activation of the MKKK by GTP binding regulatory proteins (i.e., Ras, Rac, Cdc42, Rho) or phosphorylation by another kinase (MKKKK, i.e., Mst1, PAK) controls the activity of a specific MAPK pathway. Currently, biochemical and genetic analysis has defined five different MAPK pathways in mammalian cells. The best understood include the extracellular-signal response kinase 1 and 2 (ERK1/2), ERK5, c-Jun NH2-terminal kinase (JNK) and p38 MAPK pathways. To date, 12 MAPKs, 7 MKKs, and 20 MKKKs have been identified in mammalian cells. Many of the MAPKs and MKKs are closely related kinases within the ERK, JNK, and p38 pathways.
The MKKKs are more diverse, allowing a differential control of specific MKK-MAPK component members to various cellular stimuli including cytokines, stress (i.e., heat shock, osmotic imbalance, hypoxia, drug exposure, etc.), and cell-cell contact. MKKKs also appear to be differentially expressed in different cell types; however, no detailed analysis of their expression has been published. Similarly, specific MKKs and MAPKs appear to be differentially expressed in cells of different tissue origin. The differential expression of the MKKKs in particular, but also MKKs and MAPKs, will dictate functional responses in different cell types to specific stimuli. The differential expression of MKKKs, for example, can allow one cell to respond with activation of the JNK pathway while another cell does not. To define the requirement and function of specific MAPK pathways in different cell types and in response to specific stimuli, we have used targeted gene disruption by homolgous recombination to abolish the expression of different MKKKs. These include MEKK1, 2, 3, and 4 which were originally cloned in my laboratory and selectively regulate the ERK1/2, ERK5, c-Jun kinase (JNK) or p38 pathways. More recently, we have made gene knockins to inactivate the kinase activity of specific MEKKs but the kinase-inactive protein is still expressed. In parallel with the gene knockout studies in mice, the laboratory uses live cell fluorescence imaging and fluorescence resonance energy transfer (FRET) to study the spatial and temporal activation, localization and function of MAPK pathways in cells. Combined with biochemical analysis of the proteins in MAPK signal relay systems, our studies define the role of MAPK pathways in the integrated response of cells to different stimuli and their physiological function in tissues and organs.
Journal of Biological Chemistry Cover
