Research Interests
- Antibiotic Resistance in Neisseria gonorrhoeae
- Mechanisms of chromosomally mediated antibiotic resistance
- Biological fitness consequences of acquiring resistance to cephalosporins and compensatory mutations that increase fitness
- Development of inhibitors of lipid A biosynthesis for treatment of N. gonorrhoeae
- P2Y Receptors
- Regulation of P2Y Receptor signaling in platelets
- Signaling mechanisms of P2Y receptors
Research Synopsis
Antibiotic Resistance Mechanisms
My laboratory is also interested in the mechanisms of antibiotic resistance in the pathogenic bacterium, Neisseria gonorrhoeae, the etiologic agent of the sexually transmitted infection, gonorrhoea.
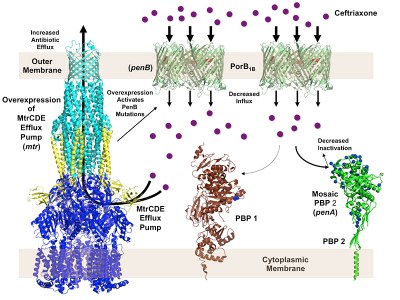
In 2012, the WHO reported over 106 million gonorrhea infections worldwide. Recently, strains resistant to ceftriaxone, the last remaining effective antibiotic, have emerged, portending an era of untreatable gonorrhea. All strains with increased and full resistance to ceftriaxone contain a highly mosaic penA gene encoding penicillin-binding protein 2 (PBP2), the lethal target of ceftriaxone, with up to 70 amino acid mutations. Other mutations include mtrR, which increases expression of an efflux pump, and penB, which encodes mutations in the major outer membrane porin, PIB (fig. 2).
We are investigating the structural mechanisms of resistance of PBP2, how the presence of a highly altered PBP2 impacts biological fitness and pathogenesis, and identifying compensatory mutations arising in the mouse model of gonococcal infection that increase fitness of mosaic penA-containing strains.
Platelet P2Y Receptor Regulation
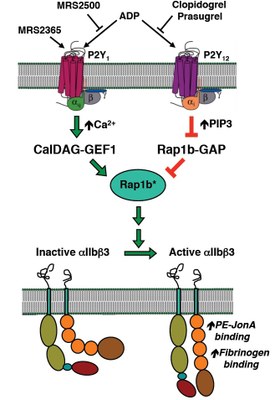
P2Y receptors are important receptors in platelet biology. When platelets come in contact with underlying tissue following disruption of the blood vessel, they release dense granules containing secondary mediators, resulting in aggregation of passing platelets and formation of a platelet plug. One of the most important of these mediators is ADP, which acts on two P2Y receptors: P2Y1 and P2Y12. The P2Y1 receptor couples to Gq, mobilizes intracellular Ca2+, and activates PKC, whereas the P2Y12 receptor couples to Gi and inhibition of adenylyl cyclase (the P2Y12receptor is the target of the anti-platelet drug Plavix). Simultaneous activation of these two receptors is required for aggregation (Fig. 1). The P2Y1 receptor is uniquely regulated and rapidly desensitizes (t1/2 of ~18 sec), preventing further aggregation. We are investigating the molecular mechanisms of this rapid desensitization in both a mouse transplantation model and knock-in mice expressing a non-desensitizing receptor mutant receptor.

