Clinical Core
About
The goal of the UNC CFAR Clinical Core is to enhance all aspects of human HIV research. We provide easy access to comprehensive clinical services, resources and expertise supporting basic science, translational, clinical and public health research.
The Clinical Core offers support with recruitment, enrollment, protocol design development and implementation. Core personnel are available to assist with regulatory and study coordination support, data and specimen collection, web-based tracking and data entry, database design and data management.
The Clinical Core maintains the UNC CFAR HIV Clinical Cohort (UCHCC), with adjudicated clinical outcomes data and samples, HIV sequences, behavioral surveys and patient reported outcomes. Ongoing collaborations exist with the CFAR Network of Integrated Clinical Systems and other national and international cohort studies. For more information visit the UCHCC website.
The Clinical Core holds regular trainings for research professionals. Please access our open source training materials here. We also hold Work in Progress presentations to give investigators the opportunity to present and receive feedback on current work. Prior presentations are available on the UCHCC website.
Members of the Clinical Core work closely with the AI in HIV Research (AIHR) Research Interest Group (RIG). The AIHR RIG promotes collaboration, training, and capacity-building, and aims to harness AI’s potential to improve diagnostics, treatment, and health outcomes for individuals living with HIV. The AIHR RIG also hosts a seminar series on the use of AI in research. For more information or inquiries about the AIHR RIG, contact us at: CFARClinicalCore@med.unc.edu.

Activities/Services
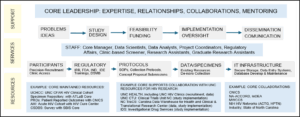
- Consultation for clarification, review, or teaching of essential clinical and clinical research concepts.
- Provision of texts and reference literature.
- Provision of online resources and reference material for investigators.
- Statistical and epidemiologic software tutorials.
- Classroom teaching and specialized lectures for Schools of Medicine and Public Health.
- Review and critique of existing / draft protocols.
- Provide customizable catalogues of SOPs, data and sample collection protocols, and regulatory applications.
- Assistance in framing of hypotheses, identification of clinical, therapeutic, metabolic, epidemiological, behavioral and disease specific factors.
- Collaborative development of study design (experimental and observational).
- Development of clinically appropriate analysis plans and strategies.
- Justification of proposed methods and strategies, or comparison of methods.
- Assistance with investigational new drug (IND) submissions.
- Budget planning for grant proposals, estimation of costs.
- Provision of accurate and up-to-date feasibility assessments through the UCHCC.
- Letters of support, biosketch, other support pages.
- Assistance with obtaining background information.
- Writing of major sections, or collaborative editing of text.
- Critiques of existing grant proposals.
- Assistance with IRB and GCRC application.
- Assistance with data collection or identification, and assessment of appropriateness to research hypotheses.
- Guidance with model choice and selection of appropriate analytic technique.
- Assistance with clinical epidemiologic analytic computations.
- Interpretive analyses of clinical, laboratory and basic science results.
- Collaborative preparation of publications and presentations.
- Consultation on design of database management plans for individual projects.
- Consultation on the operation and maintenance of project-specific systems.
- Assistance or consultation in design and creation of data collection instruments and forms, including web-based data entry systems.
- Access to UNC CFAR HIV Clinical Cohort (UCHCC) to assess subject availability and provide reliable background information.
- Access to the UCHCC for observational clinical based research.
- Database coordination with other Cores and Centers.
- Preparation of datasets suitable for statistical analysis.
- Consultation on study implementation, operations, and monitoring adherence to protocol.
- Consultation regarding communication, coordination, and timelines in large studies.
- Subject identification, recruitment and consent.
- Identification and collection of clinical samples.
- Access to stored samples from well characterized patients.
- Assistance with study design and development.
- Conducting or assisting in the conduct of clinical research trials.
- Access to expertise in multiple sub-disciplines of HIV research including oral medicine, hepatology, cardiology, neurology, nephrology, hematology/oncology, obstetrics and gynecology, pulmonology, and psychiatry.
- Access to expertise in the development and implementation of HIV vaccine trials.
- Publish and present expository papers for HIV researchers.
- Guidance and consultation to new CFAR members.
- Mentoring investigators with CFAR Developmental Awards or NIH K Awards.
- Mentoring dissertation or masters thesis students, infectious disease fellows, medical students, undergraduates, and physician extender students working on a CFAR project.
- Mentoring of international clinical and laboratory researchers.
- Monthly collaborative research meeting.
- Annual World AIDS Day HIV/AIDS symposium.
- Annual Community Forum for HIV infected patients.
- IGHID Friday Conferences
- CFAR Work in Progress Presentations
- Artificial Intelligence in HIV Research Seminars
- Special Topics in HIV Research
- Identification and recruitment of appropriate professional clinical and basic scientists available to assist new or ongoing projects.
- Successful referral and follow-up with monitoring and verification of client satisfaction.