Researchers including Greg Wang PhD at the University of North Carolina at Chapel Hill have uncovered a new mechanism that activates specific genes, leading to the development of cancers.
They showed that a mutation that fuses two unrelated genes can promote a process similar to that observed when oil and water are mixed but do not blend together. The process, called liquid-liquid phase separation, occurs inside a cell’s nucleus and enables the formation of compartments with various physical properties that can promote cancers such as acute leukemias (see Illustration). Their findings will be published online June 23, 2021, in Nature.
Publication links can be found here: Phase separation drives aberrant chromatin looping and cancer development. Nature and also here: Fusion proteins form onco-condensates. Nature Structural & Molecular Biology

“Phase separation has been a missing puzzle piece in understanding the development of human cancers,” said UNC Lineberger’s Greg Wang, PhD, associate professor of Biochemistry and Biophysics and Pharmacology at the UNC School of Medicine and co-lead author of this research article. “This finding is among the first to link phase separation to development of these terrible diseases.”
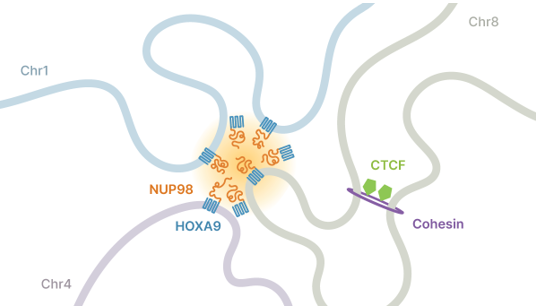
The discovery brings new insights to a complex, multistep process that bridges biology and physics. To help unravel this process, the investigators performed laboratory experiments in cancer cells that carried a common gene fusion called NUP98-HOXA9. This aberrant fusion is found solely in blood cells of patients that develop leukemia.
“Because similar gene fusions have been observed in other malignancies, the mechanism we elucidated could explain other types of cancer as well,” said Douglas H. Phanstiel, PhD, assistant professor of Cell Biology and Physiology at the UNC School of Medicine and co-lead author of this article. “We believe that our research could open up new and innovative avenues to attack cancer cells.”
Within the proteins produced by NUP98-HOXA9 are unstructured stretches, known as intrinsically disordered regions, or IDRs. The role of IDRs has also been a mystery, but the researchers showed that these IDRs promote liquid-liquid phase separation of the NUP98-HOXA9 proteins when they reach critical concentrations in the nucleus, causing NUP98-HOXA9 to become phased, or compartmentalized.
“The way that liquid-liquid phase separation alters the behavior of NUP98-HOXA9 proteins is that it makes them bind to target genes much more strongly”, said Jeong Hyun Ahn, PhD, a postdoctoral fellow at UNC and first author of this article. “DNA binding of NUP98-HOXA9 proteins, when they are phase-separated, generates a unique pattern called a super-enhancer”. Stronger, super enhancer-like binding by NUP98-HOXA9 proteins to DNA leads to more potent activity of this factor, which underlies the formation of aggressive blood cancers.
“Theoretically, a drug that specifically disrupts or dissolves the phase-separated liquid droplets formed by NUP98-HOXA9 could be a therapeutic agent,” said Wang. “We hope to look into possible therapeutic agents that target phase separation as we know that this process can also impact neurodegenerative diseases such Alzheimer’s where plaques that build up in the brain may be due, in part, to phase separation.”
The researchers also discovered that phase separation can influence the three-dimensional structure of the genome by creating structures called chromatin loops. Chromatin loops organize the genome and help control which regions are active and which are not. But alterations in this structure can cause human disease when they were aberrantly formed.
“Our discovery is the first clear evidence of chromatin loops formed by phase separation,” Phanstiel said. “This new class of loops appears to be driving cancer development by connecting regulatory regions of chromatin to cancer genes, thereby increasing cancer gene expression and lethality.”
Taken together, the complex interplay of biology, physics and genetics inside a cell is now better understood due to this latest research finding. Having performed most of their experiments in the lab, the scientists hope to be able to study certain aspects of the process in living organisms and other diseases in the near future.
Publication links can be found here: Phase separation drives aberrant chromatin looping and cancer development. Nature and also here: Fusion proteins form onco-condensates. Nature Structural & Molecular Biology.
Authors and disclosures

In addition to Wang, Phanstiel and Ahn, the paper’s other authors are Eric S. Davis, Timothy A. Daugird, Shuai Zhao, Ivana Yoseli Quiroga, Hidetaka Uryu, Jie Li, Yi-Hsuan Tsai, Daniel P. Keeley, Ling Cai and Wesley R. Legant, UNC; Aaron J. Storey, Samuel G. Mackintosh, Ricky D. Edmondson, Stephanie D. Byrum, and Alan J. Tackett at the University of Arkansas for Medical Sciences, Little Rock; and Deyou Zheng at the Albert Einstein College of Medicine, Bronx, NY.
This work was supported by NIH grants P30-CA016086, P30 NS045892, U54 HD079124, R01-CA215284, R01-CA218600, R35-GM128645, DP2GM136653, P20GM121293, R24GM137786, R01CA236209, S10OD018445, TL1TR003109, R01HL148128, R01HL153920 and T32-GM067553 as well as North Carolina Biotechnology Center Institute Development Program grant 2017-IDG-1005.
The authors declare no competing interests.
News courtesy of UNC Lineberger Comprehensive Cancer Center communications.
Open access PDF “Phase separation drives aberrant chromatin looping and cancer development”
See coverage in GEN, “Cancer Formation Linked to Phase Separation” (June 24, 2021)
See also UNC Lineberger Twitter thread.