A major route toward creating effective vaccines against dengue virus and Zika involves the E protein that covers the surface of each viral particle. If we could develop strong antibodies against this E protein, then that would be the crux of a formidable vaccine – based on the important fact that the 180 E proteins come in pairs. But creating such a vaccine has proven difficult for a number of reasons. Now UNC School of Medicine researchers have delineated the details of one major barrier to a promising vaccine. It’s something we all have – a natural body temperature of about 98.6 degrees.
Previous studies in Aravinda de Silva’s lab have shown that humans create strong antibodies to the E protein on the virus surface, suggesting that a soluble version of the E protein (called sRecE) could make a good vaccine. Unfortunately, so far this has not panned out, and vaccines based on sRecE have failed. UNC researchers led by Brian Kuhlman, PhD, professor of biochemistry and biophysics, and Aravinda de Silva, PhD, professor of microbiology and immunology, have shown that body temperature reduces the pairing propensity of sRecE and its ability to be recognized by these strong antibodies.
These findings, Published in the Journal of Biological Chemistry, suggest that stabilizing the pairing propensity of sRecE may be critical to creating an effective vaccine.
“We’ve shown how our natural body temperature subverts sRecE potency and how future sRecE-based vaccine development needs to take temperature into account from the beginning,” said UNC graduate student Stephan Kudlacek, the first author on the JBC paper.
After decades of intense research, dengue and Zika virus vaccine development has had limited success. These mosquito-borne flaviviruses are a major public health concern. More than 50 percent of the world’s population is at risk of infection, hundreds of millions of people are infected, and a large percentage of them face serious, debilitating symptoms. Dengue infection can lead to life-threatening hemorrhagic disease. Meanwhile, the recent Zika outbreak has linked Zika infection to serious neurological disorders, such as microcephaly in infants and Guillain-Barré Syndrome in adults. For these reasons, vaccine development has been ramped up to protect against both dengue and Zika.
Vaccine development is challenging because there are four different serotypes or “flavors” of dengue, and a vaccine needs to provide equal protection against each of them. Also, a vaccine has to be created such that it doesn’t promote the immune system to make antibodies that recognize but can’t neutralize the virus. If that happens, dengue and Zika can use these antibodies to infect immune system cells and make the infection more severe – this is called antibody dependent enhancement.
This inherent problem is why Dengvaxia – the only licensed vaccine for dengue – is now only recommended for use in people with partial immunity to dengue obtained from a previous dengue infection because the vaccine appears to increase the risk of severe disease when used in people who have never been exposed to the virus, and the vaccine provides partial protection between the different dengue serotypes.
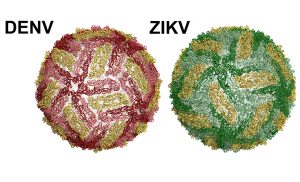
The surface of dengue and Zika viral particles are structurally similar. Both viruses are coated with a single protein – called the envelope (E) protein. Each virus contains 180 copies of the E protein on the surface, in pairs of two, known as homodimers. A soluble version of the E protein, called sRecE, can form the same homodimer structure observed by E proteins on the surface of the virus. Dengue- and Zika-infected patients develop neutralizing and protective antibodies that target specific regions on the E protein called epitopes. This knowledge has led researchers to test whether the E protein or sRecE can serve as effective vaccines. Unfortunately, sRecE-based vaccines have performed poorly.
Other researchers have discovered that many of the most potent neutralizing antibodies isolated from human patients bind to epitopes that are present on the E protein homodimer on the surface of the virus, while the weakly neutralizing antibodies that promote disease enhancement bind to a site on the E protein monomer – a single copy of the E protein, known as the fusion loop.
A particular class of the potent antibodies called the E Dimer Epitope (EDE) antibodies strikingly recognizes a region of the E protein in both dengue and Zika. These antibodies are capable of neutralizing all four dengue serotypes and Zika. These discoveries suggest that to create effective vaccines, it will be important to engineer antigens that present these newly discovered dimer epitopes.
Dimer-specific epitopes could be the most straightforward kind to create, if using recombinant proteins like sRecE. And it has been proposed that sRecE homodimers may function as effective vaccines.
But studies have suggested that our natural body temperature of 98.6 degrees changes the E protein’s ability to pair and form homodimers on the virus surface. As sRecE is a promising antigen for vaccine development, and since the stability of the sRecE homodimer is important for presenting dimer antibody epitopes such as the EDE for vaccination, Kuhlman and de Silva’s labs sought to understand how temperature affects the stability of the sRecE protein and its ability to form homodimers.

“We used a number of biophysical techniques to answer these questions.” Kudlacek said. “As typically observed, we found that colder temperatures help to keep proteins in their native or active structure. Because of this, we then tested to see how sRecE from three dengue serotypes and in Zika can form homodimers, as seen on the surface of the virus, in buffer at room temperature.”
The UNC researchers discovered that dengue and Zika sRecE do form homodimers at room temperature.
“But when we did the experiment at physiological temperature [98˚F], all sRecE’s – including dengue serotype 2 (DENV2) and Zika sRecE homodimers – fell apart and became monomers. This result showed that the physiological temperature-induced E protein changes observed on the virus surface also occur in the sRecE.”
Further analysis showed that the ability for DENV2 sRecE to form homodimers at body temperature is reduced 150 fold when compared to the ability to form homodimers at room temperature.
The researchers then wondered: since sRecE dimerization is reduced at physiological temperature, are antibodies that specifically recognize the E dimer able to bind to dengue and Zika sRecE at physiological temperature?
They tested this and found that the dengue and Zika sRecE homodimers fell apart at 98.6F, thus eliminating the dimer epitopes present in the sRecE homodimer and reducing antibody binding.
“Our results show that, at body temperature, the presence of dengue and Zika sRecE homodimers is greatly reduced, and the presence of sRecE monomer is increased,” Kudlacek said. “This provides an explanation for why previous sRecE-based vaccine strategies have performed poorly, since body temperature favors monomer sRecE, thus focusing the immune system on generating more weakly neutralizing antibodies, which can result in disease enhancement, rather than producing potently neutralizing dimer-specific antibodies.”
For future sRecE-based vaccine development, this work suggests that the sRecE homodimer needs to be re-engineered to be stable at body temperature so the immune system can generate E dimer-specific antibodies strong enough to protect against disease instead of enhancing it.
The National Institutes of Health and the Centers for Disease Control and Prevention funded this research.
Media contact: Mark Derewicz, mark.derewicz@unchealth.unc.edu, 984-974-1915